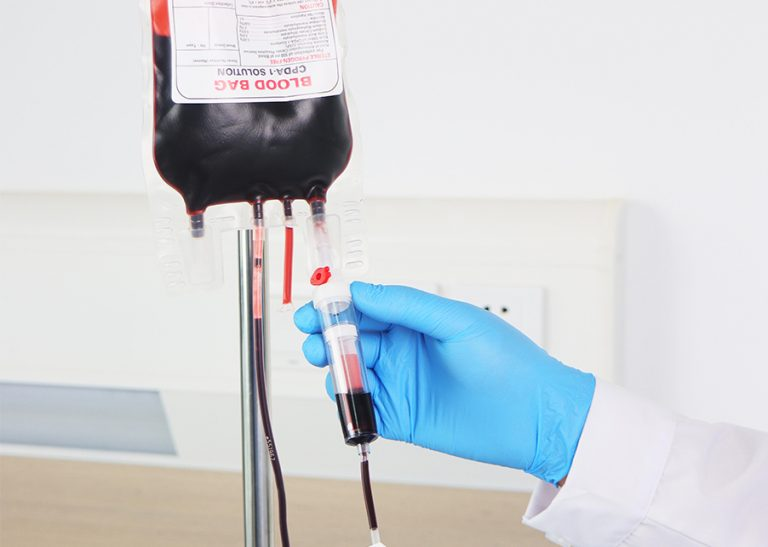
How can healthcare professionals ensure the biological integrity of a donated unit from the very first step? The process hinges on meticulous technique and reliable equipment to prevent the introduction of contaminants. We at WEGO Medical understand that the integrity of a single blood collection bags system is paramount, as any compromise can affect the safety of the blood supply. This discussion outlines a procedural framework for maintaining sterility and function during the use of these critical devices.
Pre-Collection Preparation and Inspection
Vigilance begins before the donor is even prepped. The first defensive measure involves a thorough visual and tactile inspection of the single blood collection bags unit. We advise checking the integrity of the vacuum seal, ensuring the bag is free from leaks, cracks, or any signs of prior damage. Verifying the sterility of the external port protectors is equally important. Furthermore, the work environment must be organized to have all necessary ancillary supplies—such as antiseptics, sterile gauze, and bandages—within easy reach to avoid breaking procedure mid-collection. This preparatory stage, often overlooked, establishes the foundation for a contamination-free collection when using a single blood collection bags system.
Aseptic Technique During Venipuncture
The critical moment of needle insertion presents the highest risk for microbial introduction. A strict, non-negotiable aseptic protocol must be followed. This involves effective skin disinfection of the venipuncture site with an appropriate antiseptic, applied correctly with sufficient contact time to allow it to dry completely. The technique must avoid re-palpating the cleansed site. When connecting the tubing from the single blood collection bags, care is taken to avoid contact between the needle hub and any non-sterile surface. Maintaining a closed system from the donor’s vein directly into the bag is the core principle that prevents airborne or touch contamination during the fill process.
Post-Collection Handling and Initial Processing
Once the target volume is collected, proper management of the unit continues to protect its contents. The initial mixing of the blood with the anticoagulant present in the bag must be done gently and thoroughly, following the manufacturer’s guidance to prevent clot formation. Sealing the tubing using validated methods, such as metal clips or dielectric sealers, is performed carefully to maintain the closed system. The labeled unit should then be placed in a validated temperature-controlled environment without delay to preserve component viability. Each of these steps, executed with precision, safeguards the donation until it reaches the laboratory for processing.
The goal of a successful blood collection is to deliver a unit that is as safe and potent as when it left the donor. Achieving this relies on a chain of precise actions, each link fortified by conscientious technique and dependable equipment. The design and manufacturing of our WEGO Medical collection systems are undertaken with these procedural demands in mind. By adhering to a structured protocol that emphasizes inspection, aseptic execution, and careful handling, healthcare workers can confidently use a single blood collection bags system to support a robust and secure blood supply chain.